Researchers Can Measure Distances in Molecules Optically
October 10, 2024 -- A team led by physicists Steffen Sahl and Stefan Hell at the Max Planck Institute for Multidisciplinary Sciences in Göttingen and the Max Planck Institute for Medical Research in Heidelberg has succeeded in measuring distances within biomolecules using a light microscope, down to one nanometer and with Ångström precision. The intra-molecular resolution achieved with MINFLUX microscopy makes it possible to optically record the spatial distances between subunits in macromolecules and thus to detect different conformations of individual proteins in the light microscope.
What does it look like in the nanocosmos of living cells? Fluorescence microscopy makes it possible to visualize specific molecules in cells or tissues and has become an integral part of biological and basic medical research. With the help of new high-resolution fluorescence nanoscopy concepts such as MINFLUX microscopy, even closely spaced biomolecules can be separated from each other using a light microscope. Accurately imaging the inside of cells is one thing, but can fluorescence microscopy also be used to measure details within individual proteins or other macromolecules?
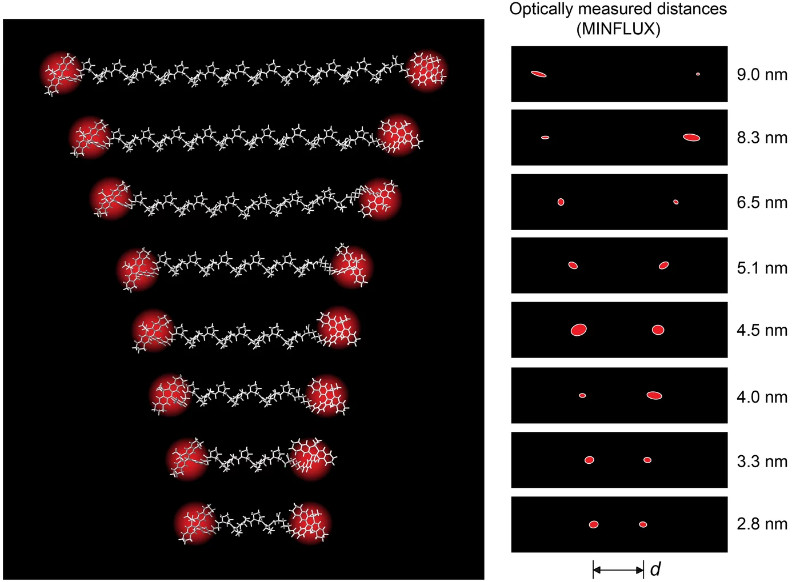
As the researchers led by Sahl and Hell have now demonstrated, it is possible. And it works remarkably well. In their latest work, the team showed that the MINFLUX method can also be used to optically measure the three-dimensional distance between two fluorescent molecular markers, each attached to a specific site of a macromolecule – and this with Ångström precision.
Measuring distances of a few nanometers between two spatially anchored fluorescent molecules was conceivable with MINFLUX, but not so easy to implement or demonstrate experimentally. “At distances of less than 5 to 10 nanometers, the fluorescent molecules, which are about 1 nanometer in size, often interact with each other. As a result, they cannot emit their fluorescence light independently of one another – the prerequisite for a reliable distance measurement,” explains Sahl, lead author of the study. “Like many others, I was fascinated by the high spatial resolution and precision that is possible with the MINFLUX method proposed and developed by Stefan Hell,” the physicist says. “At the beginning of our work there was an estimate: this is how small proteins are, and this is how precise we can become in principle. Why don't we realize resolutions within a biomolecule?" Because until now, detecting nanometer distances between two proteins, or their subunits, has been the prerogative of a method termed Förster resonance energy transfer, or FRET for short, a standard method in structural and molecular biology.
Advancing into the FRET range
Sahl, Hell and their colleagues have now also advanced into this resolution range with MINFLUX microscopy. They used photoactivatable fluorescent molecules specially developed at the Max Planck Institute for Multidisciplinary Sciences, which can be “switched on” one after the other with a small dose of UV light, but do not interact with each other. In this way, the positions to be measured in the macromolecule could be marked with a single fluorescent molecule and recorded independently with Ångström precision.
“We have shown that with MINFLUX all distances – right down to the direct contact of the fluorescent molecules – can be measured. To do this, it is sufficient to determine the positions of the molecules in two or three dimensions, that is 2D or 3D,” explains Sahl. “With our experiments, we reach the distance range of FRET and even go beyond it.”
FRET, on the other hand, estimates the distance between two dye molecules indirectly via the energy transfer from one dye to the other. Not only the distance but also the orientations of the dye molecules affect the measurement result. This can lead to uncertainties when it comes to precisely measuring the intra-molecular distance. The FRET method has also often been limited in studies of protein subunits when they move outside the measurable distance range. “This is where the MINFLUX method can demonstrate its strengths by correctly representing all conceivable distances down to 1 nanometer without any gaps,” Hell says. “MINFLUX is therefore a new, very powerful tool in the repertoire of structural biology for investigating proteins and other biomolecules and their interactions.”
Of molecular rulers and small protein molecules
To demonstrate precise distance measurement and accuracy, the research team used a molecule with which the FRET method actually started off in a classic experiment in the 1960s. At that time, Lubert Stryer and Richard Haugland managed to confirm the distance dependence published by Theodor Förster in 1948. To do this, they used molecular “rulers” of defined average length called polyprolines. The Max Planck researchers have now used exactly these rulers in their experiments – and have shown that the method can in principle even be used in cells.
In collaboration with Stefan Jakobs' research group at the Max Planck Institute for Multidisciplinary Sciences, fluorescently labeled lamin proteins in human cells, which form approximately 3-nanometer-thin filaments on the membrane around the cell nucleus, were imaged separately using the MINFLUX method. In addition, the scientists demonstrated the potential of MINFLUX through experiments with other small proteins, termed nanobodies, and their oligomers. Using antibody molecules as an example, they showed how multiple position measurements make it possible to resolve the spatial position of the protein subunits relative to one another.
Using two identical subunits of the bacterial citrate sensor, the team, in collaboration with Christian Griesinger's Department at the Max Planck Institute for Multidisciplinary Sciences, demonstrated that even distances of 1 nanometer can be measured. MINFLUX microscopy also clearly revealed the two structural arrangements of the subunits with a precision in the range of 1 Ångström.
Hell, who was awarded the Nobel Prize in Chemistry in 2014 for the development of super-resolution microscopy, is pleased: “Since we first demonstrated the MINFLUX concept in 2016, it has once again radically pushed the boundaries of light microscopy. Being able to resolve within macromolecules was not really foreseeable in 2014.”




































